What is ChemECar?
In ChemECar, we design and build a small car that is powered and controlled by chemical reactions. Our goal is to get as close as possible to a target distance, chosen on the day of competition. Think of this like ice curling, except it's focused on reaction kinetics and safety.
flag Competition Goals
- Build a shoebox-sized car
- Power the car using a homemade electrochemical cell
-
Stop the car using a chemical clock timerBe as close to the target distance as possiblegroup My group's focuses
Objective - Stopping Mechanism
Our constraints were simple: start a chemical reaction, measure its progress, and stop the car at exactly the target distance. The only variable we can control is how much reactant we put in the chemical timer. Sounds fun, huh?
Reaction Vessel and Chemistry
We've learned our lesson from prior designs (our 3D printed reaction cell was not great for containing liquids), and decided to use brass plumbing fittings for our reaction vessel.
Why brass fittings?
- Liquid Containment: These 1/4" standard pipes and fittings are designed to hold liquid, perfect for our needs.
- Low cost: No more than $60 worth of Ace Hardware components and a few sensors
- Standard Threads: standard fittings allowed us to plug in 2 pressure gauges and a pressure relief valve seamlessly.

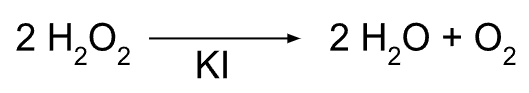
Decomposition of hydrogen peroxide via potassium iodide catalyst
We decided to use household reactants (potassium iodide, hydrogen peroxide) to perform the decomposition reaction, as they are readily available and already in safe concentrations. They also produce only oxygen and water as byproducts, making waste disposal simple.
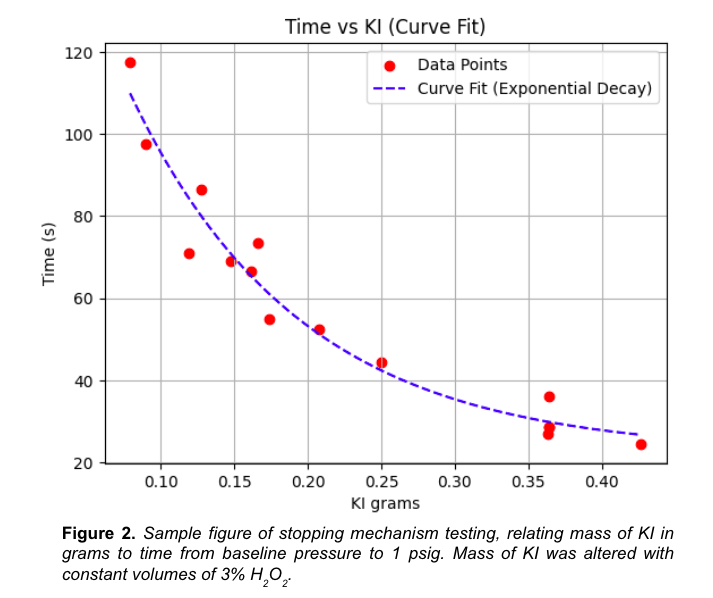
We varied the mass of catalyst added, and recorded the time until the pressure threshold was reached. After collecting our data, we had a good curve fit to model the reaction kinetics. Why is this helpful in the first place? It allows us to add exactly the needed catalyst in order for the car to run for a specific amount of time.
The chassis design team and the power cell team give us the car speed. Knowing the car speed and the distance needed, we determine exactly how long it should run for. Then we put that much catalyst in and let it run!
Engineering Challenges
One significant challenge was the ambient temperature sensitivity of the reaction kinetics. We were baffled for several weeks trying to figure out why the reaction behaved differently while we thought all variables were being controlled. We eventually realized that ambient temperature fluctuations were the cause, especially when we held the vessel during cleaning. To mitigate this, we decided to run the reaction always at a known temperature: 0 degC. We achieved this by jacketing the vessel with icewater.
Safety
Building pressure in a sealed container like this is dangerous. We installed a manual pressure gauge as a backup to the pressure sensor to make sure we are always aware of the internal pressure. We installed a pressure relief valve set at 5 psig, to make sure we are within the AIChE rules (and so we don't have to hydrotest the vessel). This was a narrow range, so we had to use a very accurate pressure sensor. We also limited how much hydrogen peroxide was added: we calculated that if all of it decomposed, it would not exceed the design pressure of the brass components.



